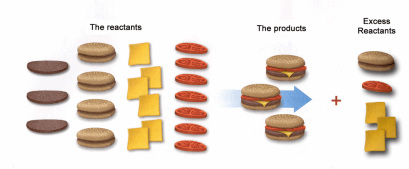
If you have these supplies, which one is limiting? Which one will you run out of first?
In this example it is easy to "see" which is the limiting and which is excess. When looking at quantities of chemicals it is not as easy to "see."
To determine which chemical is limiting, convert from the given reactants to a product (it does not matter which). Whichever reactant produces the least amount of product is limiting.